Current Research
Overview
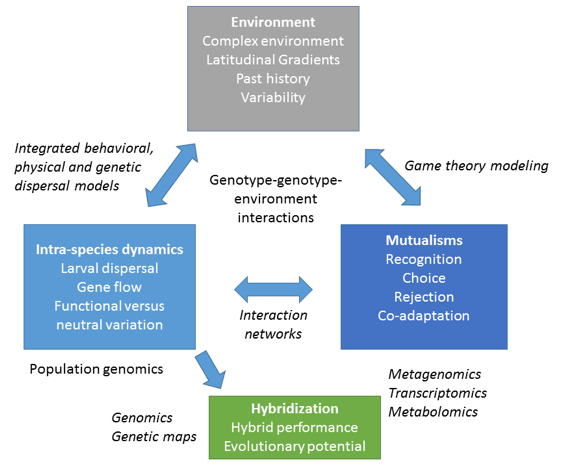
I am an evolutionary ecologist focusing on stress responses and population structure of cold water and warm water corals to understand their ability to adapt or acclimatize to environmental change. I complement field experiments with molecular techniques, including population genomics, gene expression, epigenetics and metabolomics assays. I am particularly interested in understanding the interactions between coral hosts and their symbionts under changing environmental conditions (Fig 1). My lab’s research is supported by grants from the National Science Foundation, NOAA, and NGO’s.
Evolutionary Ecology of Cold-water Corals
My laboratory integrates across the disciplines of genetics, ecology, evolution and oceanography to understand the processes that shape populations in marine environments and create highly diverse and productive systems. Deepwater coral communities comprise more species than shallow water communities do, and can have diversities exceeding those recorded on the Great Barrier Reef. They have been found in every ocean and deep-sea corals are thought to have given rise to shallow water coral lineages. Deep coral communities are exposed to natural (e.g. cold seeps) and anthropogenic oil and gas inputs. During the Deepwater Horizon oil spill, coral communities in the path of the oil plume were directly impacted. However, subacute impacts of the oil and dispersant on deep water coral are poorly understood. We conducted non-lethal oil and dispersant exposure experiments on the deep sea corals, Leiopathes glaberrima and Paramuricea sp. B3 and showed that dispersant and dispersed oil caused higher mortality and greater gene expression changes than and oil alone (1, 2). We are actively exploring the role of microbial symbionts of deep water corals in metabolizing input of cold seep derived organics such as oil and gas. Characterization of the microbial symbionts is a focus of the lab for the coming years.
Intra-species Dynamics
Starting from a population genetics background, my lab has shown that genetic connectivity among coral populations is limited despite the presence of potentially widely dispersing planktonic larvae (3-10). My paper on the lack of gene flow across the Eastern Pacific Barrier (a 5000 km stretch of open water between the central and eastern Pacific) in the coral, Porites lobata, was featured as a “perspective” in the journal Molecular Ecology (8). This paper led to an international collaboration where we used a bio-physical dispersal model to show that, in contrast to long-standing hypotheses, El Nino Southern Oscillations do not change current patterns sufficiently to allow for a breach of this barrier from east to west (11). We applied our population genetic markers to, for the first time, estimate the age of coral genets; that is we used somatic mutations to date the time when egg and sperm met to form an extant coral genet (12, 13). Surprisingly, we found that some coral genets might be several thousand years old. This begs two questions. Given the lack of a segregating germ line in some cnidarians, are somatic mutations passed on to the next generation? Do somatic mutations lead to phenotypic divergence of clone members? In the next five years, we will be investigating the role of somatic mutations in coral acclimatization and adaptation.

Inter-species Dynamics
The fate of corals is inextricably linked to their intracellular symbionts; however, co-evolution of corals and symbionts on short time-scales is poorly understood. We study how genetic structure of host and symbionts combine to alter the performance of the symbiosis. We developed markers that resolve the individual genotype/cell line of both hosts (14, 15) and symbionts (16). We found that 83% of A. palmata colonies (n = 792) harbor only one clone (cell line) of Symbiodinium ‘fitti’ (17) and, for the first time, provide evidence of sexual recombination in colonies that harbor two symbiont cell lines. We then exposed to temperature stress six common-garden reared A. palmata colonies of different genotype that share one symbiont cell line, thus allowing us to control for symbiont cell line as a contributing factor (18). Photosynthesis efficiency and response to temperature of the symbiont cell line was dependent on the host genotypic background. Similarly, host gene expression differed with temperature and the efficiency of the symbiosis. Thus, the unique combination of host and symbiont genotypes (genotype-by-genotype interaction) provides a source of variation that may serve as a foundation for thermal adaptation.

Acclimatization
Acclimatization is a non-genetic process by which an individual organism heightens its stress tolerance after exposure to a stressor, such as temperature anomalies. Recent work has shown that acclimatization may be an important process by which corals may survive climate change. However, because reef-building corals harbor intracellular symbionts (genus Symbiodinium), discerning the relative contribution of host and symbiont to acclimatization can be difficult. The endangered Caribbean elkhorn coral, Acropora palmata, has an uncomplicated symbiosis: it associates with just one symbiont species (S. ‘fitti’) and most colonies also harbor only one strain of S. ‘fitti’ over space and time (see above). August of 2014 was the warmest on record for the Florida Keys. By early September 2014, numerous corals species bleached throughout the Florida Keys reef tract. This event provides an unprecedented opportunity to understand the role of acclimatization in reef corals. Initial surveys of A. palmata documented a range of bleaching response. This response varied between reefs but also within single, monoclonal stands of A. palmata. Thus, coral clonemates growing within centimeters of each other were observed to exhibit different bleaching susceptibilities despite sharing identical (clonal) Symbiodinium populations as determined by genotyping, begging the question as to what mechanisms account for such bleaching differences. Preliminary data indicates that other members of the microbiome are also not responsible. Instead, host methylation patterns influenced bleaching phenotypes. This has prompted a new direction of research that I plan to pursue for the next five years: what role do epigenetic changes play in the ability of corals to express radically different phenotypes from the same genotype. This work is in collaboration with Sally Mackenzie (PSU) and Roberto Iglesias-Prieto (PSU), and J Lopez (FIU). We just received a new NSF grant that will fund some of this work. The answers may fundamentally shape our understanding of how reefs might survive climate change.
Hybridization - Harbinger of Extinction or Shortcut to Adaptation
Current models predict the demise of reefs in the next 200 years due to increasing sea surface temperatures and ocean acidification. It is thus essential to identify habitats, taxa and evolutionary mechanisms that will allow some coral species to maintain their role as foundation fauna. Hybridization can provide an avenue for adaptation to changing conditions. Corals hybridize with some frequency and results may range from the introduction of a few alleles via introgression, to the birth of a new, perhaps better-adapted genetic lineage. The only widely accepted coral hybrid system consists of the once dominant but now threatened Caribbean species, Acropora cervicornis and A. palmata. In the past, hybrid colonies originating from natural crosses between elkhorn and staghorn corals were rare, and evidence of hybrid reproduction was limited to infrequent matings with the staghorn coral. Our recent field observations suggest that the hybrid is increasing and its ecological role is changing. These hybrids appear to be less affected by the disease that led to the mass mortality of their parental species in recent decades. Hybrids are also thriving in shallow habitats with high temperatures and irradiance suggesting they may be less susceptible to future warming; and they are expanding into the deeper parental species habitats.
Our genetic data indicate that hybrids are now mating with each other, demonstrating the potential for the formation of a new species. Further, hybrids seem capable of mating with both staghorn and elkhorn coral, perhaps leading to gene flow between the parent species via the hybrid. Our current research addresses how the increase in hybridization and perhaps subsequent introgression will affect the current ecological role and the future evolutionary trajectory of Caribbean acroporids. Specifically, this collaborative project aims to answer the following questions: 1) what is the historic rate, direction, and degree of introgression across species ranges and genomes? Linkage block analysis based on genome-wide SNP genotyping across three replicate hybrid zones will answer this question. 2) What is the current extent and potential of later generation hybrid formation? Morphometric and genetic analyses combined with in vitro fertilization assays will be used. 3) What mechanisms allow hybrids to thrive in hot, shallow waters? A series of manipulative in situ and ex situ experiments will determine whether biotic or abiotic factors favor hybrid survival in shallow waters. We are taking a multidisciplinary approach that combines traditional and innovative technology to provide a detailed analysis of the evolutionary ecology of Caribbean corals (funded by the NSF). We just released two Acropora genomes, and I will spend the next few years leveraging these resources to further understand the potential of hybridization as a short cut to adaptation.
Laboratory Model Species
We are members of the Astrangia research consortium that is currently developing the temperate coral Astrangia poculata as a new model species. A. poculata occurs from the northern Gulf of Mexico up to Massachusetts (19, 20). It associates with Symbiodinium psygmophilum, but polyps that are very nearly symbiont-free can often be found in the same colony. Because polyps do not share a gastrovascular cavity, comparison of gene expression profiles in symbiotic and aposymbiotic portions of the same A. poculata colony reflect the difference in nutrient source of those portions. Polyps across a colony are genetically identical, eliminating background genetic variation from the host as a factor. Sexual reproduction in A. poculata occurs via spawning of gametes over several months (also in captivity), and newly settled larvae acquire symbionts via horizontal transmission. We are hoping to develop functional genomic tools (CAS-9) targeting the larval stages. Unlike other cnidarian models, A. poculata constructs an aragonite skeleton, making this species useful for reductionist and comparative studies with scleractinian corals. We have established A. poculata at PSU and it is easily cultivable far from the ocean. In vitro cultures of compatible (homologous) S. psygmophilum isolated from A. poculata are available. Using this coral, we can manipulate the symbiosis at all stages, from uptake through maintenance, and breakdown. This will allow us to investigate the extent to which cnidarians regulate symbionts through novel symbiotic pathways versus general metazoan immune components. The Baums lab is leading the effort to sequence the genome of the host and we anticipate the release of the genome by the end of 2018. Easy and cheap access to cultivable corals at PSU will be of substantial benefit for my current and future graduate students, especially in the current funding climate.

Photo Credit: Baums Lab, PSU
References cited
- D. M. DeLeo, D. V. Ruiz-Ramos, I. B. Baums, E. E. Cordes, Response of deep-water corals to oil and chemical dispersant exposure. Deep Sea Research Part II: Topical Studies in Oceanography 129, 137-147 (2016).
- D. Ruiz-Ramos, C. Fisher, I. B. Baums, Stress-response of the black coral Leiopathes glaberrima to the exposure of crude oil and dispersant Mar Pollut Bull, (subm).
- I. B. Baums, M. W. Miller, M. E. Hellberg, Regionally isolated populations of an imperiled Caribbean coral, Acropora palmata. Mol Ecol 14, 1377-1390 (2005).
- I. B. Baums, M. E. Johnson, M. K. Devlin-Durante, M. W. Miller, Host population genetic structure and zooxanthellae diversity of two reef-building coral species along the Florida Reef Tract and wider Caribbean. Coral Reefs 29, 835-842 (2010).
- N. L. Foster et al., Connectivity of Caribbean coral populations: complementary insights from empirical and modelled gene flow. Mol Ecol 21, 1143-1157 (2012).
- I. B. Baums, C. B. Paris, L. M. Cherubin, A bio-oceanographic filter to larval dispersal in a reef-building coral. Limnology and Oceanography 51, 1969-1981 (2006).
- N. R. Polato, G. T. Concepcion, R. J. Toonen, I. B. Baums, Isolation by distance across the Hawaiian Archipelago in the reef-building coral Porites lobata. Mol Ecol 19, 4661-4677 (2010).
- I. B. Baums, J. Boulay, N. R. Polato, M. E. Hellberg, No gene flow across the Eastern Pacific Barrier in the reef-building coral Porites lobata. Mol Ecol 21, 5418-5433 (2012).
- X. M. Serrano et al., Long distance dispersal and vertical gene flow in the Caribbean brooding coral Porites astreoides. Sci Rep 6, 21619 (2016).
- X. Serrano et al., Geographic differences in vertical connectivity in the Caribbean coral Montastraea cavernosa despite high levels of horizontal connectivity at shallow depths. Mol Ecol 23, 4226-4240 (2014).
- S. Wood et al., El Niño, surface circulation and coral larval dispersal across the world’s greatest marine barrier. Nat. Commun., (in press).
- M. K. Devlin-Durante, M. W. Miller, G. Caribbean Acropora Research, W. F. Precht, I. B. Baums, How old are you? Genet age estimates in a clonal animal. Mol Ecol 25, 5628-5646 (2016).
- A. Irwin et al., Age and intraspecific diversity of resilient Acropora communities in Belize. Coral Reefs 36, 1111-1120 (2017).
- I. B. Baums, M. K. Devlin-Durante, L. Brown, J. H. Pinzón, Nine novel, polymorphic microsatellite markers for the study of threatened Caribbean acroporid corals. Molecular Ecology Resources 9, 1155-1158 (2009).
- I. B. Baums, C. R. Hughes, M. H. Hellberg, Mendelian microsatellite loci for the Caribbean coral Acropora palmata. Mar Ecol Prog Ser 288, 115-127 (2005).
- J. Pinzón, M. Devlin-Durante, M. Weber, I. Baums, T. LaJeunesse, Microsatellite loci for Symbiodinium A3; (S. fitti) a common algal symbiont among Caribbean Acropora; (stony corals) and Indo-Pacific giant clams (Tridacna). Conservation Genetics Resources 3, 45-47 (2011).
- I. B. Baums, M. K. Devlin-Durante, T. C. LaJeunesse, Scales of dispersal may limit rate of co-evolution between reef corals and their dinoflagellate symbionts. Proc Natl Acad Sci, (subm).
- J. E. Parkinson, A. T. Banaszak, N. S. Altman, T. C. LaJeunesse, I. B. Baums, Intraspecific diversity among partners drives functional variation in coral symbioses. Sci Rep 5, 15667 (2015).
- T. G. Jacques, N. Marshall, M. E. Q. Pilson, Experimental ecology of the temperate scleractinian coral Astrangia danae. 2. Effect of temperature, light-intensity and symbiosis with zooxanthellae on metabolic-rate and calcification. Mar Biol 76, 135-148 (1983).
- A. Szmant-Froelich, M. Pilson, Effects of feeding frequency and symbiosis with zooxanthellae on nitrogen metabolism and respiration of the coral Astrangia danae. Mar Biol 81, 153-162 (1984).